 Overall, the periodic table with charges is useful to help determine the nature of the chemical element in its ionic form, i.e., whether it loses or gains electrons to achieve the nearest inert gas configuration. For example, helium (with atomic number 2) has a complete first shell, while in case of neon with atomic number 10, 2 electrons fill the first shell and the remaining 8 occupy the second shell, thus making the element inert. In case of inert gases or noble gases (the helium group), the electrons fill up the shell completely and their charge is zero. The common ionic charges of the elements are shown in the table below. For example, an element ‘X’ in ionic form loses 2 electrons to fill the outermost shell. If the atom loses electron, a positive charge is assigned, whereas negative sign indicates gain of electrons. So, to achieve a complete shell (nearest inert gas, neon like configuration), it donates its 1 electron, thus having a charge +1 (represented as Na 1+). List of elements with their common ionic charges. The number of charge corresponds to the number of electrons that the atom requires to have a complete outer shell. Over here, the electronic configuration is 2 electrons in the S shell, 8 in the P shell, and remaining 1 in the outer D shell. 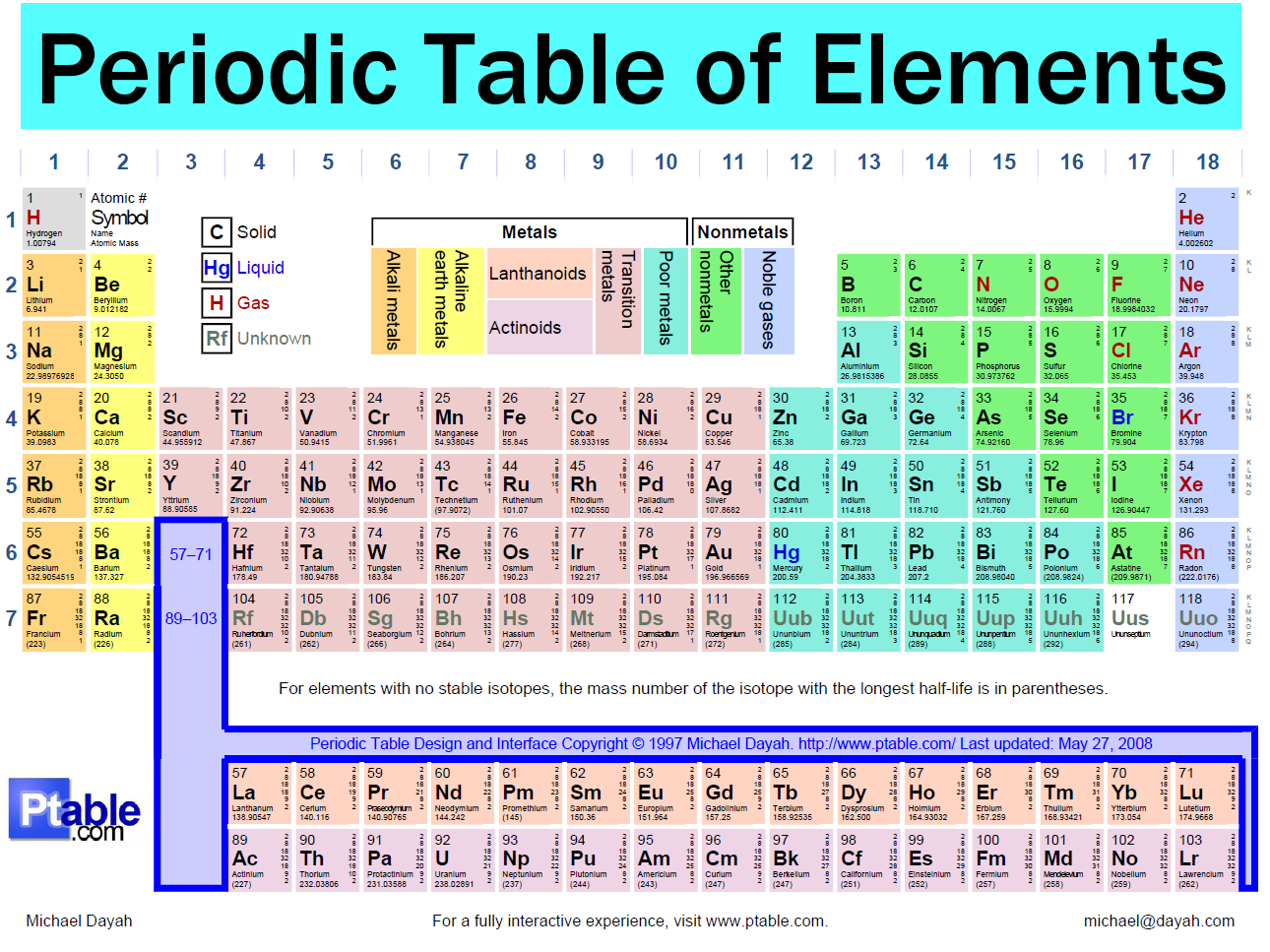
Let’s take an example of sodium (Na) that has atomic number 11. Thus, according to this method, 2 electrons are allotted in the first shell, 8 electrons in the second shell, and 18 electrons in the third shell. 
In order to find out the charge, first examine the configuration of electrons in the orbitals or shells (S, P, D, and F shells) as per the 2n 2 rule (where ‘n’ is the number of shells). Briefly speaking, the charge of an element in its ionic form refers to the actual number of electrons that it loses or gains to achieve the nearest noble gas configuration. And a periodic table with atomic mass and charges interprets both the charges and atomic mass. Likewise, the periodic table with charges illustrates elements with the probable charges, when they are in the ionic form. You may also come across periodic table with atomic mass that gives a simplified view of the elements with their respective mass numbers (total number of protons and neutrons). 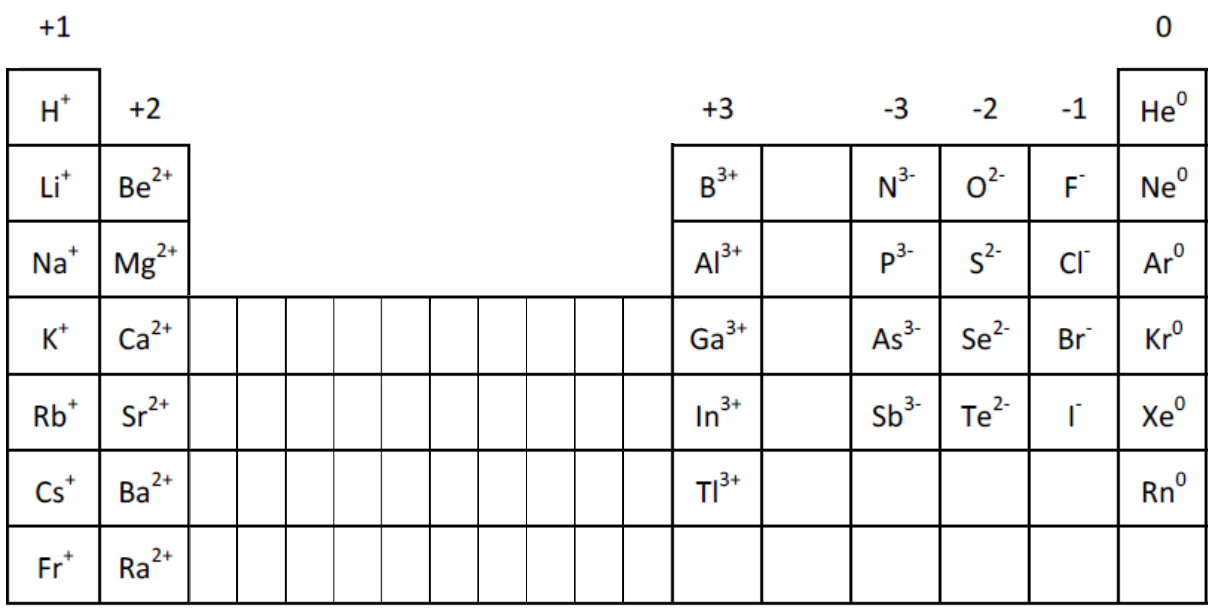
This labeled periodic table of element with names and their atomic number helps in easy interpretation of the properties of elements. Thus, instead of sorting elements as per their mass number (which is so in Mendeleev periodic table), elements are arranged from left to right, according to increasing order of the atomic number. The modern periodic table of elements, or just periodic table, is a tabular representation of the identified elements (as of date 118) along with their respective symbolic name and atomic number. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
